How the Johnson & Johnson COVID-19 vaccine works

The Johnson & Johnson (Janssen) vaccine uses a slightly different approach than the previous two COVID-19 (Pfizer and Moderna) vaccines. The Johnson & Johnson product is an adenovirus vaccine or a viral vector vaccine. Here is how it works.
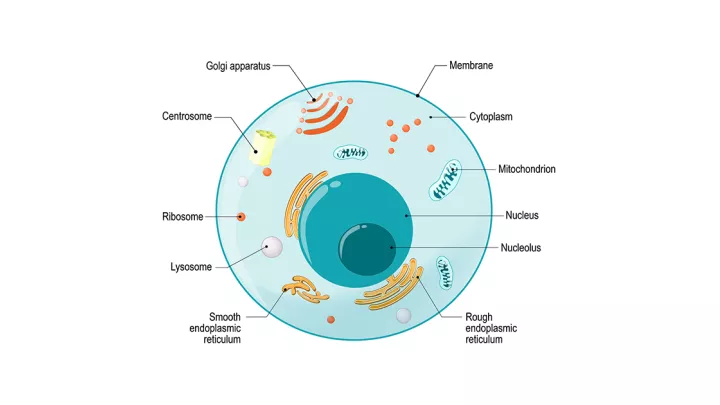
The Johnson & Johnson vaccine delivers the virus' DNA to your cells to make the spike protein. An adenovirus acts as a delivery vehicle used to carry the coronavirus genetic material (DNA). The adenovirus delivers the little piece of DNA to the cell that will then make the spike protein. After your cells produce the spike protein, your immune system creates antibodies toward the spike protein, protecting you from infection.
"Once the adenovirus gets inside your cell, the spike protein gets manufactured by your own cellular mechanisms," explains infectious diseases expert James Lawler, MD, MPH.
Usually, the adenovirus causes the common cold. But this modified virus cannot replicate, so it won't cause any disease – COVID-19, common cold or otherwise.
You are considered fully vaccinated 14 days or more after a single dose of the Johnson & Johnson shot. During that time, your immune system develops antibodies to the coronavirus spike protein. Then, if you encounter the coronavirus later, your immune system will quickly recognize it and sound the alarm.
"Part of what an antibody does is get in the way," says infectious diseases expert David Brett-Major, MD, MPH. "It prevents the virus from entering the cell, giving more time for other parts of your immune system to recognize it and attack it."
The Johnson & Johnson vaccine will not cause you to test positive to COVID-19 on a molecular or rapid protein test. It may, however, cause you to test positive on an antibody test.
How new is adenovirus technology?
Researchers have been working with adenovirus vaccines since the 1970s. Another use of the same adenovirus-based vaccine platform from Johnson & Johnson is in development against Ebola, and another against HIV. "Thousands of people around the world received this type of vaccine in clinical trials for Ebola and HIV," says Dr. Brett-Major. "One of the reasons it was approved quickly is that the safety data was already there."
AstraZeneca has also developed an adenovirus vaccine for COVID-19. It uses a different adenovirus from the one employed by Johnson & Johnson.
Also, unlike the Johnson & Johnson product which cannot replicate, the AstraZeneca replicates in the body a limited amount.
- mRNA vaccines: Moderna, Pfizer
- Adenovirus vaccines: Johnson & Johnson, AstraZeneca, Sputnik V
How J&J is different from mRNA
The Johnson & Johnson vaccine is not an mRNA vaccine (like Pfizer and Moderna). All three vaccines deliver genetic material to your cells: mRNA vaccines deliver mRNA, and Johnson & Johnson delivers DNA. Another difference is the delivery method used – either a little enclosure made of fat surrounds the genetic material, or in the case of Johnson & Johnson, an adenovirus carries it. But the end result is the same: both the mRNA vaccines and the Johnson & Johnson vaccine help your body to develop antibodies against the coronavirus spike protein.